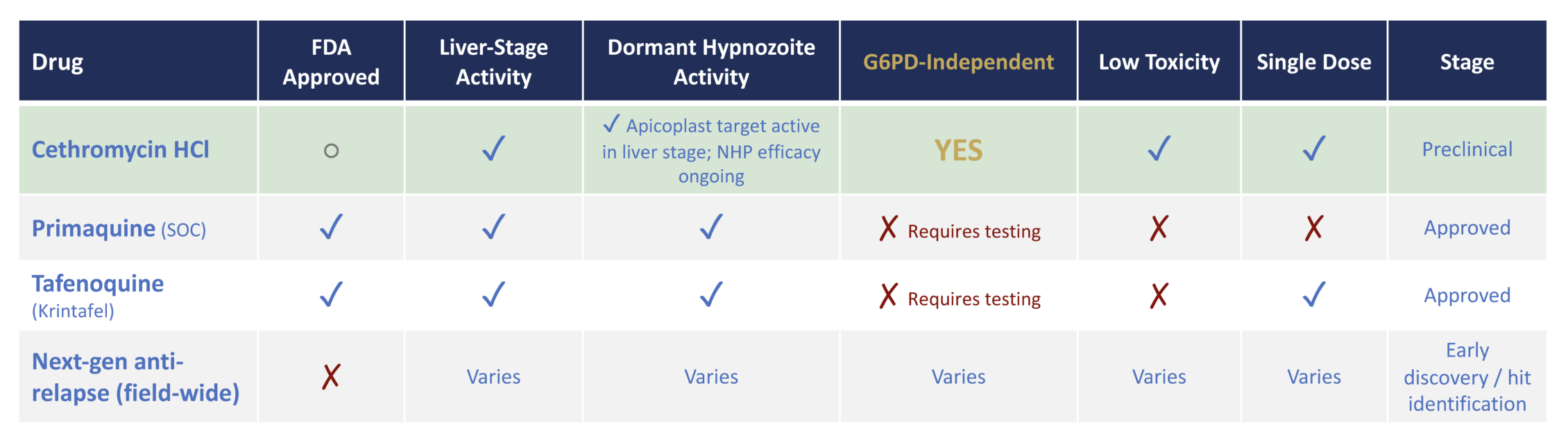
A short-course liver-stage therapy designed to eliminate dormant malaria without blood testing.
Plasmodium vivax and Plasmodium ovale form dormant liver-stage parasites that can reactivate weeks or months after initial infection. These relapses sustain transmission and disease burden even in regions with effective blood-stage control.
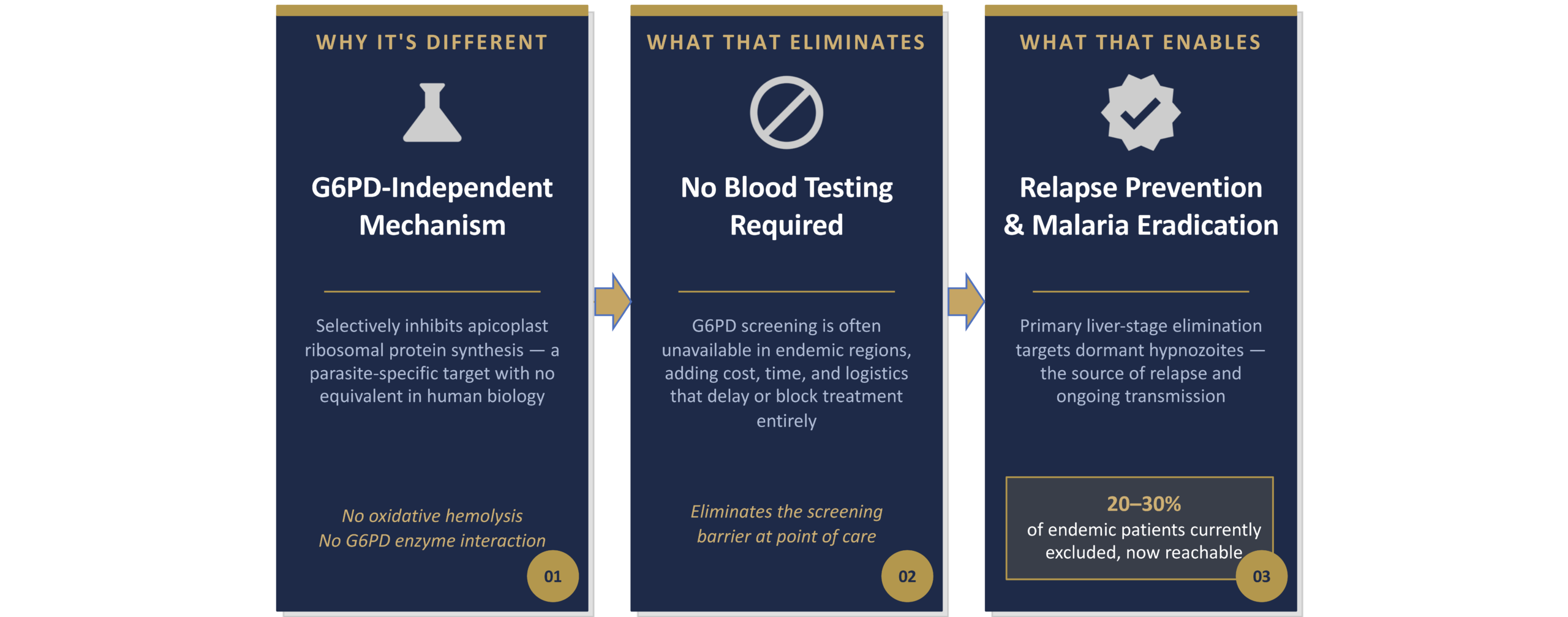
Current radical-cure regimens require G6PD testing due to the risk of hemolysis, creating a major deployment barrier in endemic settings where testing is unavailable or impractical.
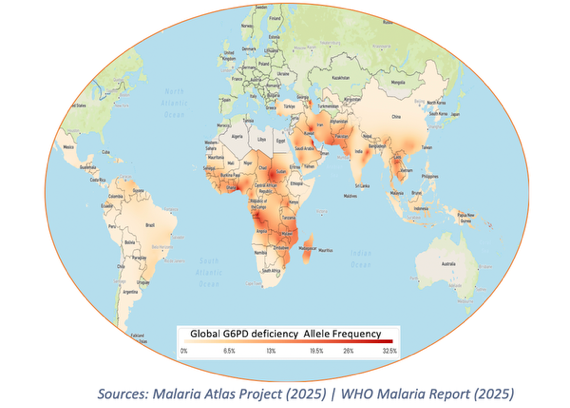
As a result:
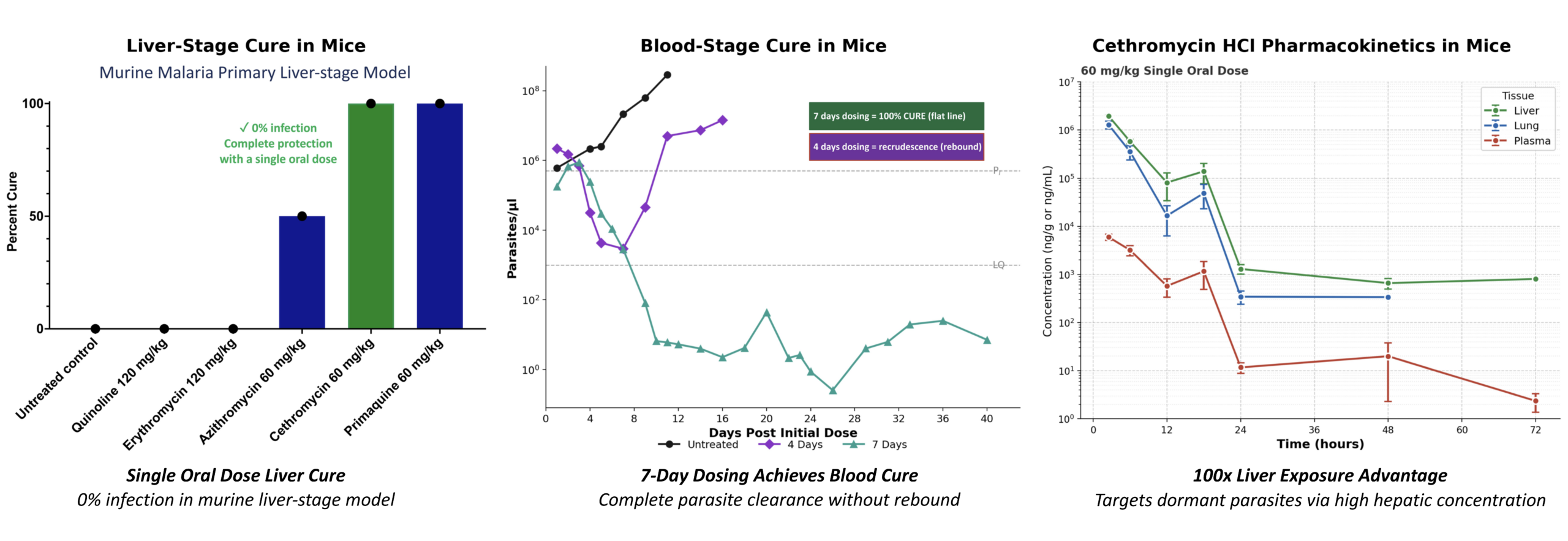
Cethromycin HCl harnesses a well-characterized macrolide mechanism validated across bacterial and parasitic systems. Its preferential liver accumulation enables sustained exposure at the site of liver-stage infection, differentiating it from traditional antimalarial approaches. Preclinical studies demonstrate liver-stage efficacy consistent with this exposure profile.
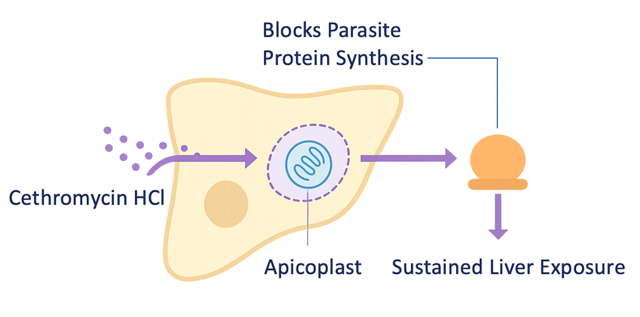
Cethromycin HCl preferentially accumulates in hepatocytes, where it inhibits apicoplast protein synthesis required for Plasmodium maturation, enabling sustained liver-stage activity.
Following FDA Type B meeting feedback in March 2026, AliquantumRx is advancing CALM on an accelerated development timeline.
Key Milestones
Q3 2026 – NHP efficacy readout
Q4 2026 – IND submission planned
Late 2026 – Phase I clinical initiation target
2027 – Phase IIa CHMI proof-of-biology study
FDA has indicated that Phase I may proceed without additional IND-enabling toxicology studies based on prior human exposure data with cethromycin base.