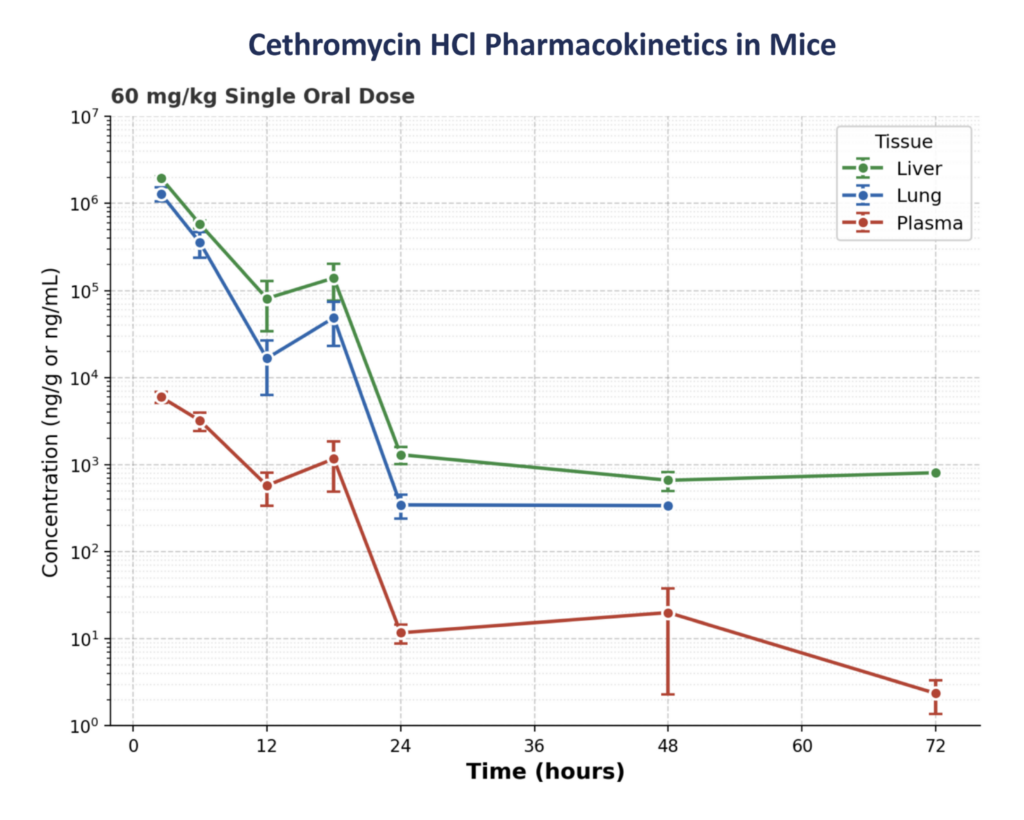
Cethromycin HCl accumulates preferentially in the liver, where dormant malaria parasites reside.
Its pharmacologic profile supports:
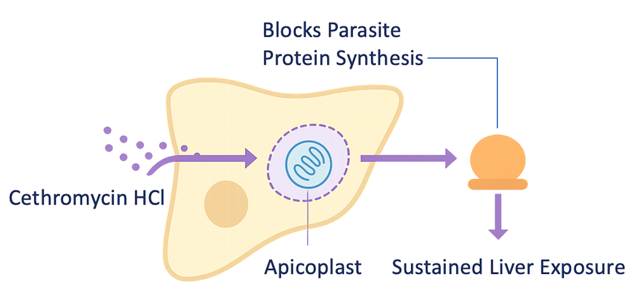
Cethromycin HCl inhibits parasite protein synthesis through apicoplast targeting — a biologically validated pathway relevant to liver-stage malaria.
Cethromycin free base has been administered to more than 5,000 human subjects in prior clinical development, establishing a substantial human exposure and safety database. This human exposure supports early clinical development and helps de-risk initial clinical evaluation.