AliquantumRx’s pipeline is initially focused on advancing its lead candidate, Cethromycin HCl, toward human proof-of-concept in liver-stage malaria relapse prevention through a malaria-first development strategy.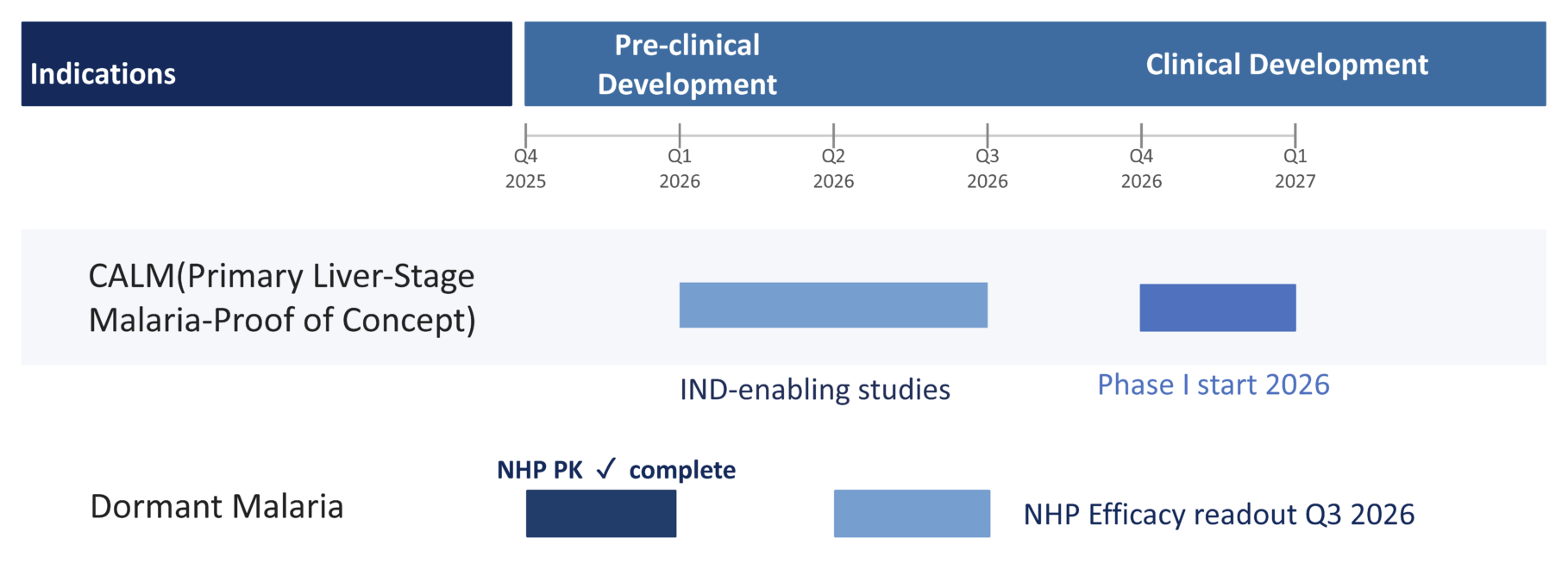
Cethromycin HCl is being developed as a short-course, G6PD-independent therapy designed to eliminate liver-stage malaria parasites and prevent relapse.
Based on its intended indication and development pathway, Cethromycin HCl may be eligible for established U.S. regulatory programs including Fast Track and Breakthrough Therapy designation, Priority Review , Tropical Disease Priority Review Voucher (PRV), Orphan Drug Act incentives, and Qualified Infectious Disease Product (QIDP) designation, subject to regulatory criteria.
Additional indications are supported by historical data and ongoing scientific collaborations, including select intracellular infectious diseases and biodefense-related pathogens; however, AliquantumRx is initially focused on achieving human proof-of-concept in liver-stage malaria.
